Eli Lilly’s Fight Against Counterfeit Medications
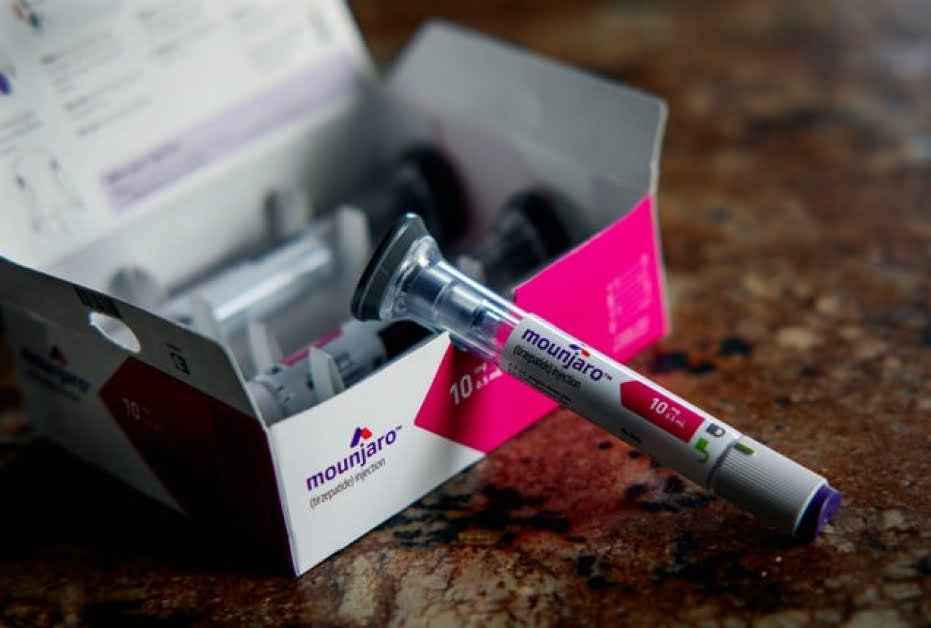
Eli Lilly, a leading pharmaceutical company, is taking decisive action against entities selling unauthorized versions of its popular weight loss and diabetes drug, tirzepatide, known by the brand names Mounjaro and Zepbound. This crackdown on counterfeit medications underscores the serious risks associated with fake drugs and emphasizes the importance of buying pharmaceuticals from trusted sources. As Eli Lilly strives to uphold the quality of its products and protect the well-being of its customers, it is crucial for consumers to be vigilant when purchasing weight loss treatments and to seek advice from healthcare professionals before starting any new medication regimen.
Legal Action Against Counterfeiters
Eli Lilly recently announced legal actions against several businesses, including med-spas and wellness centers, for promoting fake versions of tirzepatide. These entities were falsely claiming that their products were genuine Mounjaro or Zepbound, misleading patients by suggesting that their concoctions were part of Eli Lilly’s authorized clinical trials and FDA approvals. The company also issued a public warning about the dangers of using these untested products.
Rising Demand for GLP-1 Medications
Tirzepatide, along with other GLP-1 medications like Novo Nordisk’s Ozempic, belongs to a class of drugs used for diabetes and obesity management. The demand for appetite-suppressing pills has surged in recent years, with sales of Mounjaro and Zepbound reaching $5.3 billion in 2023 and continuing to rise. This increased demand has led to ongoing shortages, prompting Eli Lilly to invest billions in new facilities to meet the market needs.
Concerns About Counterfeit and Compounded Medications
Due to limited supply and high prices of GLP-1 drugs, some patients have turned to cheaper counterfeit or compounded alternatives. Eli Lilly cautions against using unauthorized products, as they may lack proper dosages, contain harmful substances, or have different chemical compositions than approved medications. Compounded drugs, which are customized by pharmacists or doctors for individual patients, are subject to strict regulations under the Food, Drug, and Cosmetic Act.
The Risks of Counterfeit and Compounded Drugs
Eli Lilly has identified safety and efficacy issues with some compounded tirzepatide products, including contamination, impurities, color variations, and structural differences from the company’s FDA-approved medications. These discrepancies pose serious health risks to consumers, as compounded drugs may not adhere to the same quality standards as regulated pharmaceuticals. It is essential for individuals to be cautious and informed when seeking medical treatments and to prioritize their health and safety above all else.